 Inertsil ODS-4 Analytical Columns, Particle Size 3µm HP, I.D.Pinnacle DB Cyano Columns (USP L10), Particle Size 5 μm, Length 30 mm, ID 2.1 mm, Restek (CAT#: STEM-C-1189-LC). 
InertSustain C18 Packed Guard Columns, Particle Size 3µm, I.D. Possible causes, Suggested Remedy Connectors not compatible with each other.If you are unsure of the compatibility of your chemical, we. Inertsil ODS-3 Analytical Columns, Particle Size 5µm, I.D. affect the compatibility of the fluid with the given pump.Optimized for capture and intermediate protein. Suitable for screening of selectivity, binding and elution conditions, and small-scale purifications. 20 to 290/310☌, Restek (CAT#: STEM-C-0527-LC) HiTrap Capto S chromatography column is prepacked with a strong cation exchange modern resin for small scale protein purification. Rtx-200 Columns (Fused Silica), Length 30 m, ID 0.32 mm, df 1.00 μm, Temp.Nucleic acids Nucleic acids can bind strongly to anion exchangers. MabSelect PrismA with its alkalistabilized protein Aderived ligand, was designed to withstand the use of up to 1.0 M sodium hydroxide solution as cleaning agent. 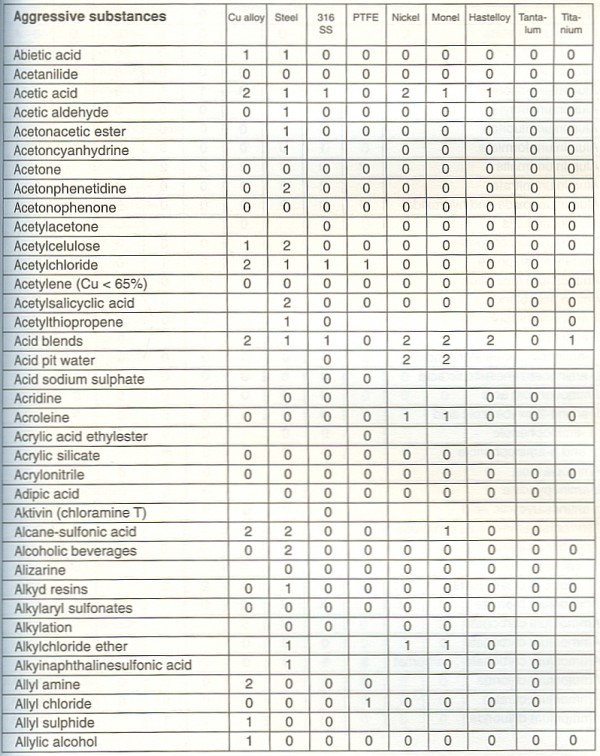
Viva C4 Columns (USP L26), Particle Size 5 μm, Length 100 mm, ID 3.0 mm, Restek (CAT#: STEM-C-1293-LC) purification, have significantly improved compatibility with sodium hydroxide.Inertsil ODS-SP Analytical Columns, Particle Size 3µm, I.D.Inertsil ODS-3 Guard Columns For UHPLC, Replacement Cartridge (2/pk) & Holder Set For UHPLC, Particle Size 3µm, I.D.The resins are used in many applications, including the purification of: Plasmids and viral vectors for gene therapy Recombinant proteins Monoclonal antibodies (mAb), Fab antibodies, and bispecific antibodies (bsAb). Clearly and legibly label each container and storage location to indicate its compatibility group. The high chemical stability of Capto resins lets you perform cleaning-in-place procedures with ease. This method enabled > 90 % overall recovery of unformulated DS at ≥ 150 g/L. Store chemical groups below separately from one another, either in separate cabinets or in appropriate tubs or secondary containers. Most of the rinse was combined with the retentate to hit the target protein concentration. 
After the retentate was collected, a minimal volume of buffer was added for the UF rinse. Figure 2 above shows the possible combinations of bed height and operational nominal flow velocity for Capto L. Choose bed height and operating flow velocity in terms of residence time, pressure restrictions and large-scale column packing challenges. During the UF/DF process, the antibody was initially concentrated to 90 g/L, diafiltered, and concentrated to ≥ 180 g/L, then the retentate was collected. Fig 2.Operating window for Capto L (white area). Analysis showed that the Capto S run removed the excipients with yields of ≥ 96%. High binding capacity, > 100 mg mAb/mL resin. In addition, Capto S has lower resin costs, takes less time to process, and uses milder elution conditions. With a high binding capacity and small particle size, Capto S ImpAct is a good choice for reliable and robust polishing steps in an industrial purification process. The high flow agarose matrix provides particle rigidity without. Please note that this does not work for polycarbonate membranes because these membranes have more than one refractive index (1.62 and 1.58). 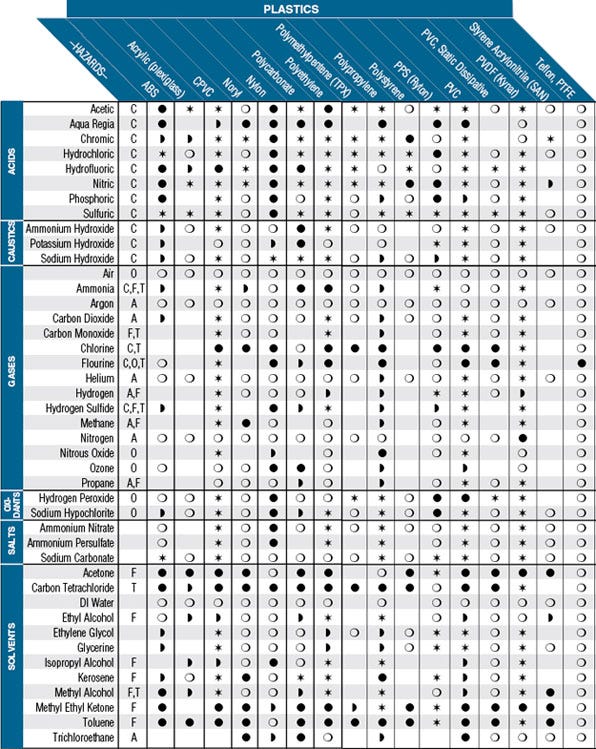
The refractive index for Durapore membrane is 1.42 and for MF membrane its 1.50. Capto adhere, Capto MMC Capto Q, Capto S MabSelect Xtra MabSelect SuRe Capto Core Sepharose 4 Fast Flow Sepharose High Performance Sepharose 6 Fast Flow Sepharose Big Beads 0 20 40 60 80 100 120 140 160 180 200 4 KA7685111119AN Fig 4. A Capto S column was chosen over Protein A chromatography to remove excipients from formulated drug substance because of its higher binding capacity. The ligands, Q, S and DEAE, are coupled to a chemically modified, high flow agarose matrix. Capto S is a strong cation exchange bioprocessing medium, designed to meet the demands of modern large-scale manufacturers for fast, efficient, and cost-effective capture and intermediate protein purification when high volume throughput is essential. This technique can be used for most membrane filters with a single refractive index. Fortunately, a sufficient supply of formulated DS was available for reprocessing. Since the pilot plants were not available for large-scale campaigns, a creative alternative was needed to produce 2 kg of antibody from formulated DS for these studies. During process scale-up, the project team decided to make a high-concentration mAb drug substance for subcutaneous injection and change the formulation. The scope of this work is two-fold: 1) excipients removal from formulated mAb drug substance by Capto S chromatography and 2) UF/DF process development to make high-concentration drug substance (DS) for subcutaneous injection. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
